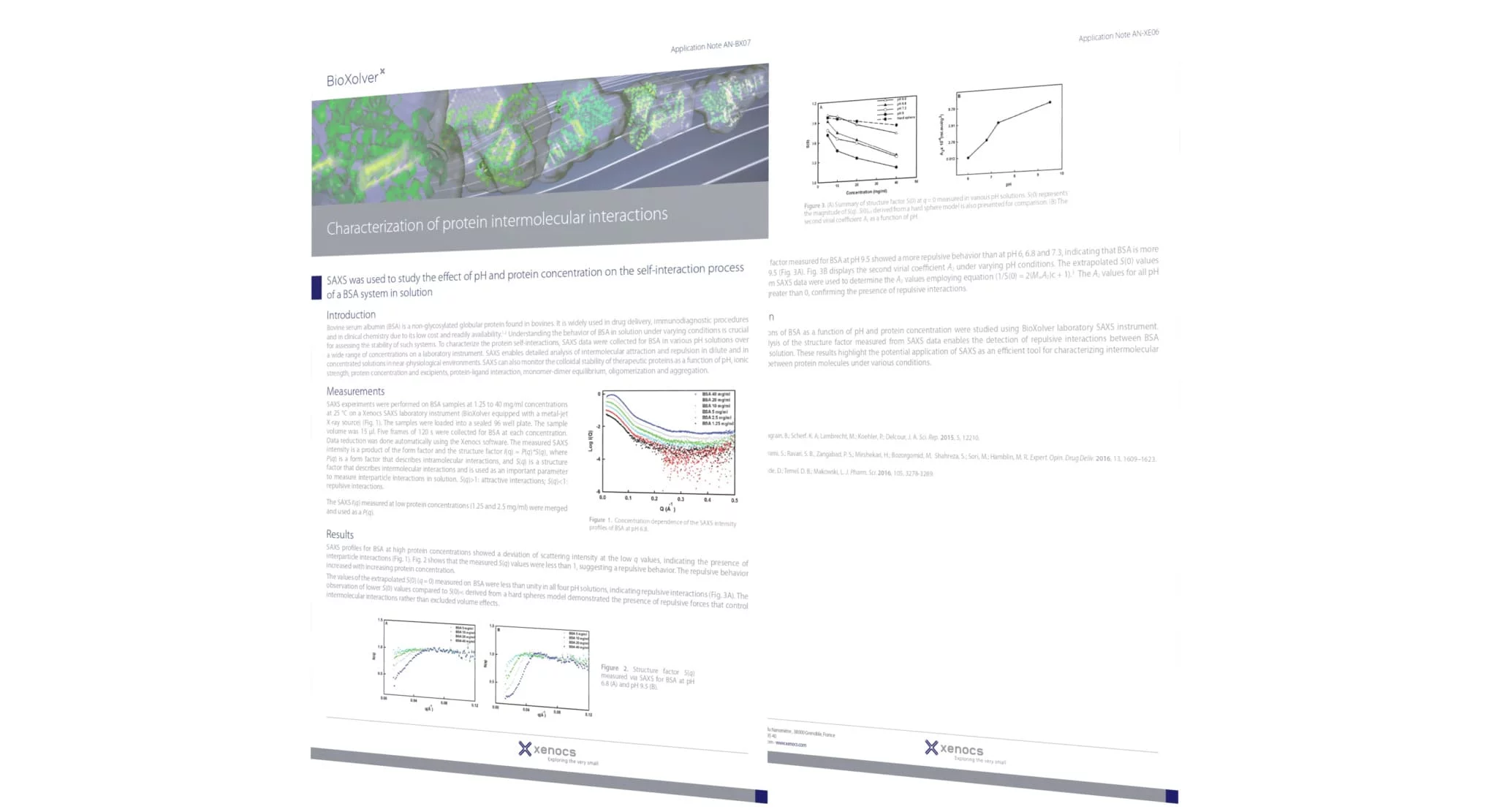
SAXS was used to study the effect of pH and protein concentration on the self-interaction process of a BSA system in solution
Bovine serum albumin (BSA) is a non-glycosylated globular protein found in bovines. It is widely used in drug delivery, immunodiagnostic procedures and in clinical chemistry due to its low cost and readily availability. [1,2]
Understanding the behavior of BSA in solution under varying conditions is crucial for assessing the stability of such systems.
To characterize the protein self-interactions, SAXS data were collected for BSA in various pH solutions over a wide range of concentrations on a laboratory instrument.
SAXS enables detailed analysis of intermolecular attraction and repulsion in dilute and in concentrated solutions in near-physiological environments.
SAXS can also monitor the colloidal stability of therapeutic proteins as a function of pH, ionic strength, protein concentration and excipients, protein-ligand interaction, monomer-dimer equilibrium, oligomerization and aggregation.