Challenges in the self-assembly of ultrahigh molecular weight block copolymers
Block copolymers (BCPs) are a special class of materials that feature two or more chemically distinct monomer units grouped into discrete polymer blocks which in turn are covalently linked together. In the melted phase, these materials exhibit microphase separation caused by the thermodynamic incompatibility between the constituent blocks [1]. This results in the formation of periodic nanomaterials (four common structures are shown in Figure 1) whose morphology can be controlled by altering the molecular composition while the size and periodicity of their domains are dictated by variations in the molecular weight [3]. Their structural and compositional versatility offers the possibility of attaining a great variety of surface nanostructures employable in an extensive number of applications such as nano-electronics, antireflective coatings, optically active surfaces chemical sensors or drug delivery [4].
Fig. 1. Four basic copolymer structures. Adapted from [2].
For optoelectronic applications employing visible light, BCPs with lateral periodicity larger than 150 nm are needed. Thus, a new sub-class of materials has emerged namely the ultrahigh molecular weight (UHMW) block copolymers. The highly entangled nature of the long polymer chains forming these BCPs however poses additional problems in the self-assembly process. Most notably the slow onset of phase separation makes nearly all processes unsuited for industrial applications. Recently a group of researchers from the University of Dublin, the University of Bordeaux and the University of Sheffield have proposed a new method of ultrafast self-assembly of UHMW BCPs (800 kg/mol) employing controlled swelling kinetics during the solvent vapor annealing (SVA) phase thus obtaining a reduction of the annealing time to minutes in contrast with the usual timeframe of hours or days [3]. In their work, they demonstrate that by rapidly and controllably swelling the film to very high levels of solvent concentration it is possible to induce the phase-separation of UHMW poly(styrene)-b-poly-2-vinylpyridine (PS-b-P2VP) systems in as short timescales as 10 minutes. To reach this result an extensive study has examined the effects of the dry film thickness, the solvent concentration inside the polymer film and the swelling time and rate on the morphology and structural evolution of BCP films.
GISAXS measurements reveal the influence of solvent concentration on the morphology of UHMW block copolymers
The long polymer chains of systems with high molecular weight display a higher entanglement of the chains in the dry films. Knowing that the polymer mobility of UHMW BCP is highly dependent on the swelling ratio, it is possible to combat this issue by adding a relatively neutral solvent to the BCP film during the SVA process. This way the molecules of the solvent will generate a screening effect at the interface between the two blocks thus reducing the polymer-polymer interactions [3]. In the aforementioned study, a mixture of chloroform and tetrahydrofuran (THF) was chosen as the annealing solvent.
Grazing-incidence small-angle X-ray scattering (GISAXS) has subsequently been used to study the evolution of the morphology of a 166 nm BCP film over macroscopic areas as a function of varying solvent concentration (ϕs). In contrast with SAXS experiments where measurements are performed in transmission mode, using a grazing mode (with the X-ray beam reflected by the sample surface) transforms the technique into a surface-sensitive probe analyzing the structure of the material over large surface areas with no additional sample preparation required. As can be observed in Figure 1 a clear evolution of the internal structure with increasing solvent concentration was detected through GISAXS measurements. The as-cast sample exhibits only weak scattering pointing to a mainly disordered film with micellar structures at the surface. Upon increasing the solvent concentration, it is evident from the GISAXS scattering patterns that below the threshold value of ϕs~0.80 the BCP chains remain too entangled to self-assemble into well-defined microdomains. It is only at concentrations equal to or higher than 0.80 that a well-ordered perpendicular lamellar morphology begins to form and evolve. Using the position of the scattering peaks, the in-plane domain spacing of the structure is calculated to be ~184 nm at ϕs = 0.83 and ~191 nm at ϕs = 0.86, while a loss of structural ordering is observed once the value of the solvent concentration reaches the value of 0.88.
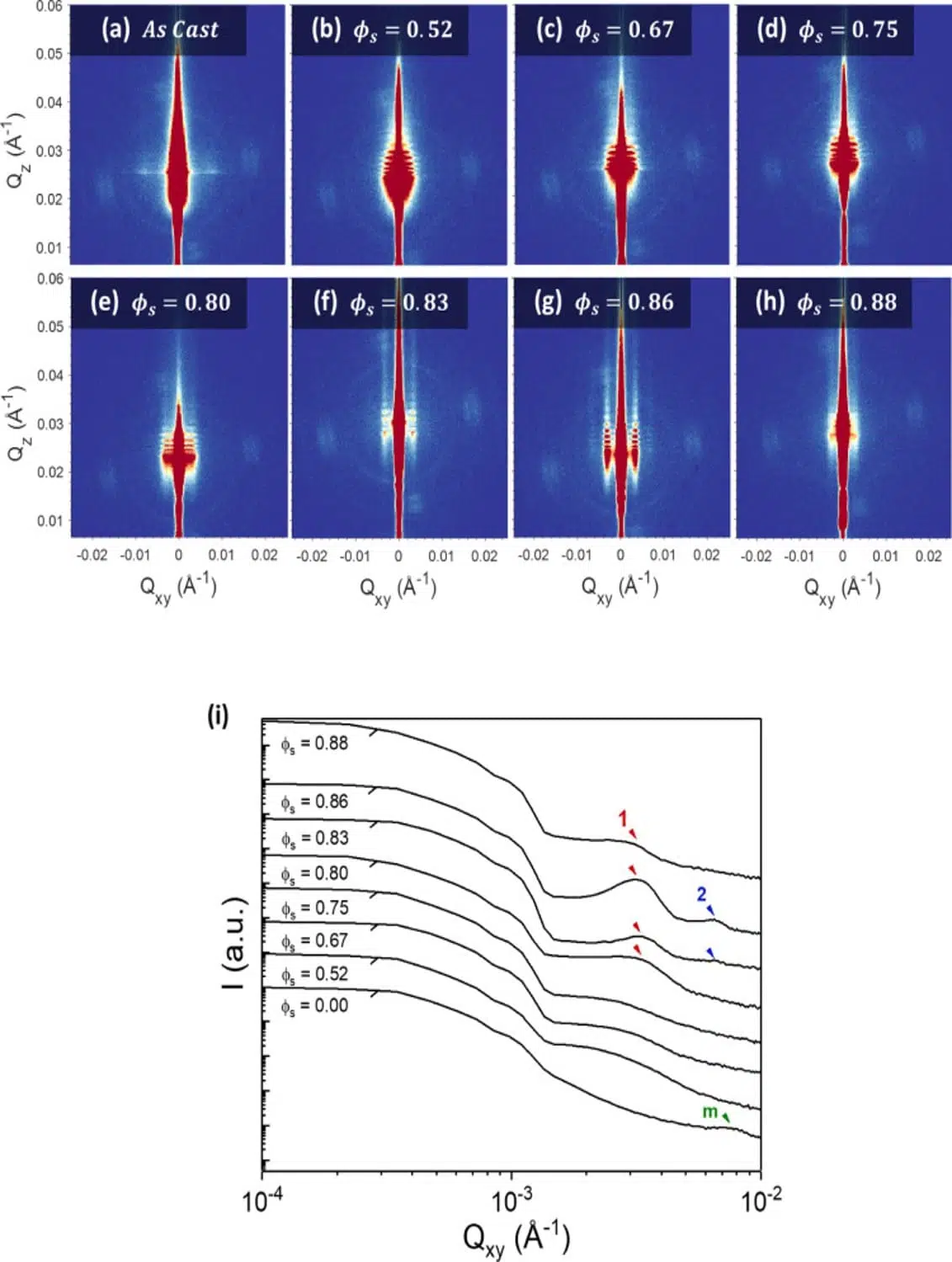
Fig. 2 (a–h) GISAXS scattering data displayed as 2-dimensional plots. The 8 panels exhibit the morphological evolution of the PS-b-P2VP films as a function of varying annealing solvent concentration ϕs. (i) 1D cuts through the GISAXS images at the deduced Yoneda position of each sample. The intensity profiles exhibit first-order, second-order scattering peaks are marked as 1 and 2 in red and blue respectively. The as-cast film (measured in the absence of solvent) displays a weak scattering peak marked as m in green. Credit: Macromolecules, 2021, DOI: 10.1021/acs.macromol.0c02543
These values are further confirmed by AFM analysis and the extension of the lamellar structure throughout the bulk of the film is proven by cross-sectional FIB/SEM measurements. To demonstrate BCP pattern transfer capability, the self-assembled films have been used as templates for manufacturing metal oxide nanostructures. These have been further employed as hard masks to produce uniform high aspect ratio Si nanowalls structures (500 nm height and 190 nm spacing).
This work underpins the feasibility of the self-assembly of ultrahigh molecular weight block copolymers through high precision vapor phase annealing in industrially applicable timeframes. Phase separation on time scales of ~10 minutes was achieved resulting in lamellar features with spacings of over 190 nm. Throughout the process, GISAXS measurements, in conjunction with other probing techniques, have been used to control the efficiency of the process and to evaluate the influence of different parameters.
































